|
8/9/2023 0 Comments Bf3 dipole moment The bond angle between the two beryllium-fluorine bonds in a beryllium fluoride molecule is 180o. NCERT Exemplar Class 11 Chemistry Solutions Chapter 2 Structure of Atom.NCERT solutions for Class 11 Chemistry Chapter 2 Structure of Atom.NCERT notes Class 11 Chemistry Chapter 2 Structure of Atom.The bond dipole moment in chemistry can be used to depict the movement of electrons. When two atoms with different electronegativities collide, the electrons tend to migrate away from their original locations in order to approach the more electronegative atom. The arrows used to illustrate dipole moments in chemistry start at the positive charge and stop at the negative charge. The bond dipole moment (µ) is a vector quantity with the same direction as the bond axis. □ → magnitude of the partial charges □ + and □ –, In a chemical bond between two atoms with differing electronegativities, the bond dipole moment formula can be written as, Where, C stands for Coulomb and m for metre. It is measured in Debye units, which are represented by the letter ‘D.' Mathematically, dipole moment formula can be written as,ĭipole Moment=ChargeQ*Distance of separation(d) The change in electron density of the molecule is shown by this arrow.īy the dipole moment definition, the dipole moment formula is the product of magnitude of charge and the distance between the positive and negative charge centres. On the positive centre, there is a cross, and on the negative centre, there is an arrowhead. The dipole moment is represented by a modified arrow sign. It can also be 0 because the two oppositely acting bond dipoles can cancel each other out as a vector quantity.Ī tiny arrow with its tail on the negative centre and its head on the positive centre is used to represent it. It's a vector quantity, which means it has both magnitude and defined directions. The bond dipole moment is the dipole moment of a single bond in a polyatomic molecule, which is distinct from the molecule's overall dipole moment. They are separated by a set distance, which is commonly symbolised by the letter 'd.'įrom the points given below we can easily understand what is dipole moment. So, what is dipole moment in case of HCl? The answer can be seen from the figure below i.e., the dipole moment that occurs in an HCl (hydrochloric acid) molecule is, The letters ẟ + and ẟ – denote the two electric charges that originate in a molecule, which are equal in magnitude but have opposing signs. It has both magnitude and direction i.e., bond dipole moment can be used to say that it is a vector quantity. It makes use of the concept of electric dipole moment, which is a measurement of the distance between negative and positive charges in a system. The moment depends on the charge at the end of the dipole and its distance from the charge at the other end of the dipole.īond dipole moment can be used to measure the polarity of a chemical bond that present between 2 atoms in a molecule of a compound. To determine if a molecule has a permanent dipole moment (and hence experiences dipole-dipole forces) you need to be able to draw a Lewis electron-dot structure for the molecule and apply VSEPR theory to determine the shape of the molecule.A dipole moment is the turning force a fixed charge has on a dipole in a molecule. For example, in the molecule BF 3, each BF bond has a dipole moment with a partial negative charge on the fluorine (it is much more electronegative than the boron), but the shape of the molecule (trigonal planar) is such that the bond dipole moments add up to zero. These bond dipole moments will be large if the bonded atoms have very different electronegativities (see this WebElements page for a graph of electronegativity across the periodic table). You can think of each chemical bond in a molecule as having a dipole moment oriented parallel to the bond, with a size and direction determined by the atoms at each end of the bond. So it is the shape of a molecule that is important in determining whether it has a dipole moment.Ī dipole moment is a vector quantity - it has both size and direction. 
This happens in molecules with sufficiently symmetric shape. The mathematical definition of the dipole moment involves adding up the size of each charge in the molecule multiplied by the average distance that charge is from an arbitrary origin.Īny molecule where the overall centre of the positive charges and the overall centre of the negative charges coincide will have zero dipole moment. 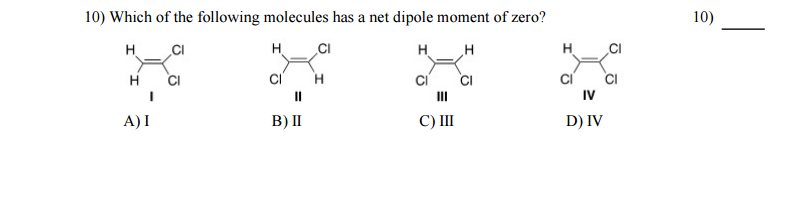
The dipole moment is a measure of the unevenness, or lack of symmetry, of the charge distribution in a molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
